
NEWSLETTER
ISSUE
Issue Jan to Mar, 2026 Volume 24
BRAIN WAVE
The Science and Strength of H₂O₂ Vapor Sterilization: A Modern Sterility Powerhouse
In an era where healthcare and pharmaceutical technologies are rapidly evolving, the need for safer, faster, and material-friendly sterilization methods has never been more pressing. Vaporized Hydrogen Peroxide (VHP/VH₂O₂) sterilization has risen as a leading solution — combining robust microbial inactivation with low-temperature operation, excellent compatibility, and strong regulatory backing.
This article explores the core principles, advantages, GMP compliance, and competitive edge of VH₂O₂ sterilization, drawing on current scientific and industrial trends.
Core Mechanism of H₂O₂ Vapor Sterilization
Vaporized Hydrogen Peroxide (VHP/VH₂O₂) sterilization works through oxidative free-radical chemistry. Using 35% concentrated lab-grade H₂O₂, ultra-pure vapor (>95% purity) is generated inside a deep vacuum chamber. Precise injections are monitored and controlled by a spectrometer to prevent condensation and ensure consistent delivery.
The vapor produces hydroxyl radicals (•OH) and reactive oxygen species (atomic O, superoxide) that attack multiple microbial cell components simultaneously — DNA/RNA, proteins, and lipid membranes.
This multi-target oxidative assault achieves a 6-log reduction of resistant spores (Geobacillus stearothermophilus) at low temperature while leaving no toxic residues (H₂O + O₂). The vapor phase enables thorough penetration into complex geometries, narrow lumens, and hard-to-reach surfaces that traditional liquid or high temperature methods cannot effectively reach.
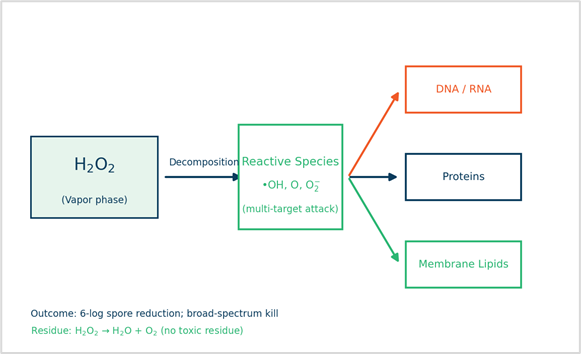
Figure 1. VHP free‑radical mechanism: catalytic/surface activation → ROS generation → multi‑target oxidative damage (DNA/RNA, proteins, membrane lipids).
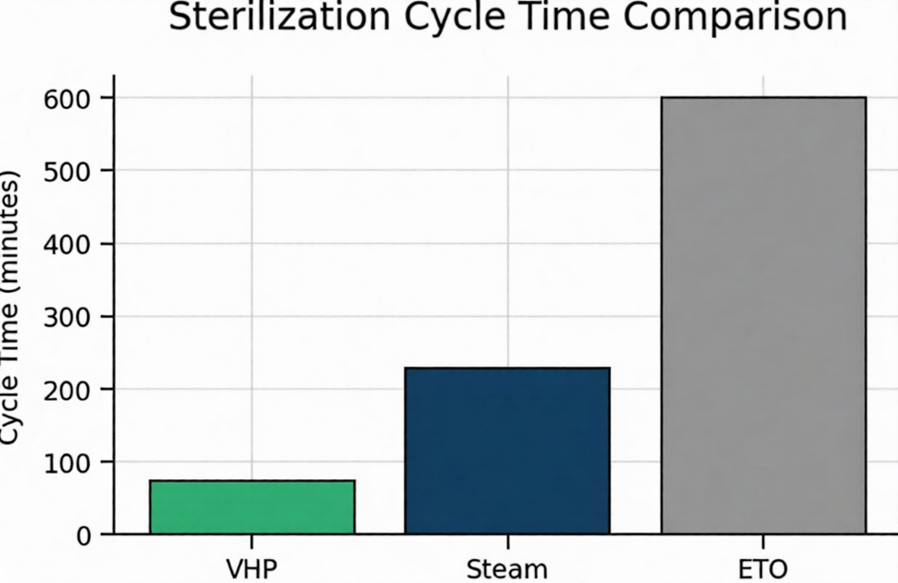
Figure 2. Typical cycle‑time comparison across modalities (illustrative).
Advantages Over Autoclaving
Steam autoclaving is limited by high temperature (121 °C+) and moisture, damaging heat- and moisture-sensitive devices, polymers, optics, and electronics. VH₂O₂ operates as true cold sterilization (40–55 °C) and competes directly with ETO, radiation, and plasma—with a clear distinction over steam ETO and plasma systems to avoid confusion: unlike steam ETO, which still relies on high heat and moisture like autoclaving (leading to similar material damage risks), VH₂O₂ delivers pure low-temperature vapor without those constraints; and compared to H₂O₂ gas-plasma, it provides 3 times more load capacity, ¼ the electricity consumption, and roughly half the capital cost. Load capacity is 15 times greater than ETO chambers, with cycle times of 35–75 minutes versus hours for ETO aeration.
Additional benefits include negligible operational and maintenance costs, reduced footprint (same size chamber delivers 3 times throughput), real-time spectrometer monitoring, standalone fully automatic UI with 21 CFR Part 11 compliance, and one-click PDF report generation.
Material compatibility & Eco-Friendliness
VH₂O₂ provides strong material compatibility across metals, polymers, plastics, electronic components, and narrow-lumen devices with no discoloration or degradation. Predefined recipes support fast, repeatable cycles. Residue management is complete — hydrogen peroxide decomposes fully into water and oxygen, eliminating toxic residues and aeration delays. As green technology, it generates zero waste and zero hazard, significantly reducing carbon footprint through minimal electric power requirements while remaining operator- and environment-friendly.
GMP Alignment & Validation Strengths
VH₂O₂ technology aligns seamlessly with GMP standards. It complies with ISO 13485:2016 (Quality Management System for Medical Devices), ISO 22441:2022 (the primary international standard for VHP sterilization of medical devices), and ISO 14937 (general requirements for validation and routine control). It also holds FDA Category A status, having been reclassified as an established, reliable mainstream sterilization process.
Validation frameworks are mature and well-established, using Process Challenge Devices (PCDs), Biological Indicators (BIs), SAL 10⁻⁶ demonstration, resistance testing, and routine monitoring with BIs and chemical indicators. Material compatibility screening (avoid cellulose; verify polymers) and sterility checks post-incubation overcome typical challenges with full confidence.
Why Switch to H₂O₂?
Traditional methods bring critical challenges — environmental hazards, material contamination, carcinogenic agents (ETO), high utility costs, and maintenance burdens. VH₂O₂ directly overcomes these issues with zero toxic residues, no carcinogenic risk, lower costs, and full compatibility with modern heat-sensitive medical and pharmaceutical products. The FDA’s endorsement of VH₂O₂ as an established Category A method confirms its status as a safe, sustainable, and future-ready alternative.
VH₂O₂ represents a balanced blend of safety, speed, material respect, and regulatory strength. It is the future-proof sterilization solution for today’s demanding healthcare and pharmaceutical environments — delivering purity without compromise.
Subscribe to our Newsletter
Stay tuned with Industry updates
| Thank you for Signing Up |

